A model system comes of age
In the paper “Symbiotic partnerships and their chemical interactions in the leafcutter ants (Hymenoptera: Formicidae)” published 2018 in Myrmecol. News, Sarah F. Worsley, Tabitha M. Innocent, Daniel Heine, J. Colin Murrel, Douglas W. Yu, Barrie Wilkinson, Matthew I. Hutchings, Jacobus J. Boomsma, and Neil A. Holmes revise the recent progress in understanding the interactions of leafcutter ants and mutualistic and parasitc symbionts. Here, Ted Schultz highlights the main points of the paper and comments on some details.
A Review compiled by Ted R. Schultz 
One-hundred and forty-four years have transpired since Thomas Belt (in Nicaragua) and Fritz Müller (in southern Brazil) independently discovered that “attine” ants cultivate fungi for food (Belt 1874, Müller 1874). Today, attine ants, their cultivated fungi, and their more recently discovered multiple microbial partners have become a model system for the study of symbiotic evolution. Progress over the past 144 years has not proceeded at a constant rate, however. Instead, it has been sporadic, with major leaps forward driven by a variety of factors. One of the most important of those factors has been, arguably, the advent of new technologies.
The earliest technology-driven discoveries occurred at the very beginning of attinology, in the iconic work of Fritz Müller’s nephew, Alfred Möller. Alfred’s mentor was the mycologist Julius Oscar Brefeld, who in 1872 invented the method of isolating and culturing microorganisms on nutrient media (in the earliest case gelatin, but soon thereafter agarose). In 1890, funded by a postdoc from the Berlin Academy of Sciences, Alfred joined his Uncle Fritz in Blumenau, Brazil, where he spent the next three years employing a variety of methods, including nutrient-media culture, to study the fungal cultivars of ten fungus-farming ant species. In his resulting monograph – the seminal work of attinology – Möller (1893) describes and illustrates the micromorphology of ant-cultivated fungi and of two parasitic fungal species that almost a century later would be given the name Escovopsis (see Muchovej & Della Lucia 1990, Seifert & al. 1995).

Acromyrmex ant colony. © Matthew I. Hutchings
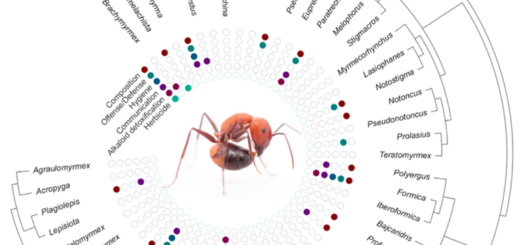
More recently, new technologies have driven major attinological discoveries in molecular ecology, molecular phylogenetics, genetics, and, most notably for our subject here, chemical ecology. Early studies in the 1960s and 1970s by the pioneering chemical ecologist Michael Martin identified three proteases in the fecal droplets of two leaf-cutting ant species and famously demonstrated that all three originated not in the ants but in their fungal cultivars, to be vectored by the ants where most needed in the garden (summarized in Martin 1987). As reviewed by Worsley & al. (2018: p. 61), during the past decade, proteomic research has increased this number to seven proteases, of which six originate in the fungus, as well as 26 other proteins (Schiøtt & al. 2010, De Fine Licht & al. 2013, 2014, Kooij & al. 2014). And now that we have genomes and/or transcriptomes of multiple symbiont species, modern attinologists have identified hundreds of enzymes and dozens of antibiotics (anti-bacterial, anti-fungal, and even anti-malarial) in attine ants, in their fungal cultivars, in the crop disease Escovopsis, and in mutualistic Pseudonocardia and Streptomyces bacterial species (e.g., Aylward & al. 2013, De Man & al. 2016, Holmes & al. 2016, Nygaard & al. 2016). Just as exciting, it is becoming increasingly possible to study and characterize chemical interactions between microbial symbionts in situ and in culture (e.g., Schoenian & al. 2011, Birnbaum & Gerardo 2016). All of this suggests that, due to the availability of new technologies as well as to attinology’s model-system status, important new discoveries are being made with increasing frequency.
I find it particularly heartening that modern attinologists are increasingly able to interpret their discoveries with reference to phylogeny, which facilitates the reconstruction of the evolutionary-historical sequences of events that produced each of the extant, interdependent clusters of symbiotic species. Thanks to genomics, we have known since 2011, for example, that Atta cephalotes lacks the genes necessary for synthesizing the amino acid arginine. It was only when we had parallel data from other species, however, that we realized that this loss likely dates to the origin of fungus-farming 55 – 60 million years ago (Ješovnik & al. 2016, Nygaard & al. 2016). Likewise, explicitly comparative in vitro bioassays of host (attine fungal cultivars) and parasite (Escovopsis spp.) interactions indicate that phylogenetic distance predicts infection success at both broad and fine scales, i.e., that Escovopsis species are generally better at infecting their native hosts or their hosts’ close relatives than they are at infecting distantly related hosts (Birnbaum & Gerardo 2016).

Sarah Worsley excavating an Acromyrmex colony. © Matthew I. Hutchings
Although it is not the explicit intent of their review, the larger message of Worsley & al. (2018) is that attinology has matured into a field in which broad theoretical questions are being simultaneously explored from multiple perspectives by a healthy number of research groups using diverse technologies. Chemical ecology is only one of a variety of disciplines that are, in an increasingly synergistic effort, exploring broad general questions about symbiotic evolution in the attine model system. With 144 years of momentum behind it, progress in attinology may still proceed sporadically, but, if the past two decades are any indication, significant leaps forward will occur with increasing frequency.
Finally, there are a few statements in Worsley & al. (2018) that require clarification. First, gongylidia, swollen hyphal tips produced by the fungus and preferentially eaten by the ants for food, are consistently produced in higher-attine fungi and usually considered absent in lower-attine fungi. However, gongylidia-like hyphal-tip swellings have been reported, albeit rarely, in lower-attine cultivars (Möller 1893, Urich 1895, Weber 1972, Mueller & al. 2001, Schultz & al. 2005, Masiulionis 2014). Whether these structures are homologous with and perhaps the evolutionary precursors of higher-attine gongylida remains a tantalizing research question. Second, the obligately symbiotic, polyploid, higher-attine fungi consist of two sister clades, one of which is represented by the single species Leucoagaricus gongylophorus and the other of which contains multiple (possibly dozens of) species (Ješovnik & al. 2017). Most leaf-cutting ants (genera Atta and Acromyrmex) cultivate L. gongylophorus (Silva-Pinhati & al. 2004), but a minority of leaf-cutters cultivate non-L. gongylophorus, higher-attine fungal species from its speciose sister clade. Likewise, most non-leaf-cutting higher-attine ants (genera Trachymyrmex, Sericomyrmex, and Xerolitor) cultivate non-L. gongylophorus higher-attine fungi, but a minority can be found cultivating L. gongylophorus (Seal & al. 2014, Mueller & al. 2018; S.Solomon, J.Sosa-Calvo, C.T. Lopes, H.L. Vasconcelos, M. Bacci, Jr., C. Rabeling, U.G. Mueller & T.R. Schultz, unpubl.). Finally, for ant-morphology sticklers only, the “laterocervical plates” mentioned by Worsley & al. (2018), which in many species of attine ants contain the crypts and glands that house and nourish actinomycete bacteria, are properly referred to as “propleural plates” or “propleurites” (Bolton 1994: p. 198, Fisher & Bolton 2016: p. 478).

Lab colony fungus garden chamber – top view. © Matthew I. Hutchings
References
Aylward, F. O., K. E. Burnum-Johnson, S. G. Tringe, C. Teiling, D. M. Tremmel, J. A. Moeller, J. J. Scott, K. W. Barry, P. D. Piehowski, C. D. Nicora, S. A. Malfatti, M. E. Monroe, S. O. Purvine, L. A. Goodwin, R. D. Smith, G. M. Weinstock, N. M. Gerardo, G. Suen, M. S. Lipton & C. R. Currie. 2013: Leucoagaricus gongylophorus produces diverse enzymes for the degradation of recalcitrant plant polymers in leaf-cutter ant fungus gardens. – Applied and Environmental Microbiology 79: 3770-3778.
Belt, T. 1874: The naturalist in Nicaragua. – John Murray, London, 403 pp.
Birnbaum, S.L. & Gerardo, N.M. 2016: Patterns of specificity of the pathogen Escovopsis across the fungus-growing ant symbiosis. – The American Naturalist 188: 52-65.
Bolton, B. 1994: Identification guide to the ant genera of the world. – Harvard University Press, Cambridge, MA, 222 pp.
De Fine Licht, H.H., Schiøtt, M., Rogowska-Wrzesinska, A., Nygaard, S., Roepstorff, P. & Boomsma, J.J. 2013: Laccase detoxification mediates the nutritional alliance between leaf-cutting ants and fungus-garden symbionts. – Proceedings of the National Academy of Sciences of the United States of America 110: 583-587.
De Fine Licht, H.H., Boomsma, J.J. & Tunlid, A. 2014: Symbiotic adaptations in the fungal cultivar of leaf-cutting ants. – Nature Communications 5: art. 5675.
de Man, T.J. B., Stajich, J.E., Kubicek, C.P., Teiling, C., Chenthamara, K., Atanasova, L., Druzhinina, I.S., Levenkova, N., Birnbaum, S.S.L., Barribeau, S.M., Bozick, B.A., Suen, G., Currie, C.R. & Gerardo, N.M. 2016: Small genome of the fungus Escovopsis weberi, a specialized disease agent of ant agriculture. – Proceedings of the National Academy of Sciences of the United States of America 113: 3567-3572.
Fisher, B.L. & Bolton, B. 2016: Ants of Africa and Madagascar. – University of California Press, Oakland, CA, 503 pp.
Holmes, N.A., Innocent, T.M., Heine, D., Bassam, M.A., Sarah F. Worsley, S.F., Felix Trottmann, F., Patrick, E.H., Yu, D.W., Murrell, J.C., Schiøtt, M., Wilkinson, B., Boomsma, J.J. & Hutchings, M.I. 2016: Genome analysis of two Pseudonocardia phylotypes associated with Acromyrmex leafcutter ants reveals their biosynthetic potential. – Frontiers in Microbiology 7: art. 2073.
Ješovnik, A., González, V.L. & Schultz, T.R. 2016: Phylogenomics and divergence dating of fungus-farming ants (Hymenoptera: Formicidae) of the genera Sericomyrmex and Apterostigma. – Public Library of Science One 11: art. e0151059.
Ješovnik, A., Sosa-Calvo, J., Lloyd, M.W., Branstetter, M.G., Fernández, F. & Schultz, T.R. 2017: Phylogenomic species delimitation and host-symbiont coevolution in the fungus-farming ant genus Sericomyrmex Mayr (Hymenoptera: Formicidae): Ultraconserved elements (UCEs) resolve a recent radiation. – Systematic Entomology 42: 523-542.
Kooij, P.W., Rogowska-Wrzesinska, A., Hoffmann, D., Roepstorff, P., Boomsma, J.J. & Schiøtt, M. 2014: Leucoagaricus gongylophorus uses leaf-cutting ants to vector proteolytic enzymes towards new plant substrate. – The ISME Journal 8: 1032-1040.
Martin, M. 1987: Invertebrate-microbial interactions: ingested fungal enzymes in arthropod biology. – Cornell University Press, Ithaca, NY, 148 pp.
Masiulionis, V.E., Rabeling, C., De Fine Licht, H.H., Schultz, T.R., Bacci, Jr., M., Santos Bezerra, C.M. & Pagnocca, F.C. 2014: A Brazilian population of the asexual fungus-growing ant Mycocepurus smithii (Formicidae, Myrmicinae, Attini) cultivates fungal symbionts with gongylidia-like structures. – Public Library of Science One 9: art. e103800.
Muchovej, J.J. & Della Lucia, T.M.C. 1990: Escovopsis, a new genus from leaf cutting ant nests to replace Phialocladus nomen invalidum. – Mycotaxon 37: 191-195.
Mueller, U.G., Schultz, T.R., Currie, C.R., Adams, R.M.M. & Malloch, D. 2001: The origin of the attine ant-fungus mutualism. – Quarterly Review of Biology 76: 169-197.
Mueller, U.G., Kardish, M.R., Ishak, H.M., Wright, A.M., Solomon, S.E., Bruschi S.M., Carlson, A.L. & Bacci, M. 2018: Phylogenetic patterns of ant–fungus associations indicate that farming strategies, not only a superior fungal cultivar, explain the ecological success of leafcutter ants. – Molecular Ecology 27: 2414-2434.
Müller, F. 1874: The habits of various insects (letter to Charles Darwin). – Nature 10: 102-103.
Möller, A. 1893: Die Pilzgärten einiger Südamerikanischer Ameisen. – Botanische Mitheilungen aus den Tropen 6: 1-142.
Nygaard, S., Hu, H., Li, C., Schiøtt, M., Chen, Z., Yang, Z., Xie, Q., Ma, C., Deng, Y., Dikow, R.B., Rabeling, C., Nash, D.R., Wcislo, W.T., Brady, S.G., Schultz, T.R., Zhang, G. & Boomsma, J.J. 2016: Reciprocal genomic evolution in the ant–fungus agricultural symbiosis. – Nature Communications 7: art. 12233.
Schiøtt, M., Rogowska-Wrzesinska, A., Roepstorff, P. & Boomsma, J.J. 2010: Leaf-cutting ant fungi produce cell wall degrading pectinase complexes reminiscent of phytopathogenic fungi. – BioMed Central Biology 8: art. 156.
Schoenian, I., Spiteller,M., Ghaste, M., Wirth, R., Herz, H. & Spiteller, D. 2011: Chemical basis of the synergism and antagonism in microbial communities in the nests of leaf-cutting ants. – Proceedings of the National Academy of Sciences of the United States of America 108: 1955-1960.
Schultz, T.R., Mueller, U.G., Currie, C.R. & Rehner, S.A. 2005: Reciprocal illumination: a comparison of agriculture in humans and in fungus-growing ants. In: Vega, F.E. & Blackwell, M. (Eds.): Insect–fungal associations: ecology and evolution. – Oxford University Press, New York, NY, pp. 149-190.
Seal, J.N., Schiott, M. & Mueller, U.G. 2014: Ant-fungus species combinations engineer physiological activity of fungus gardens. – Journal of Experimental Biology 217: 2540-2547.
Seifert, K.A, Samson, R.A. & Chapela, I.H.: 1995: Escovopsis aspergilloides, a rediscovered hyphomycete from leaf-cutting ant nests. – Mycologia 87: 407-413.
Silva-Pinhati, A.C.O., Bacci, Jr., M., G. Hinkle, G., Sogin, M.L., Pagnocca, F.C., Martins, V.G., Bueno, O.C. & Hebling, M.J.A. 2004: Low variation in ribosomal DNA and internal transcribed spacers of the symbiotic fungi of leaf-cutting ants (Attini: Formicidae). – Brazilian Journal of Medical and Biological Research 37: 1463-1472.
Urich, FW. 1895: Notes on some fungus-growing ants of Trinidad. – Journal of the Trinidad Field Naturalists’ Club 2: 175-182.
Weber, N. 1972: Gardening ants: the attines. – Memoirs of the American Philosophical Society 92: 1-146.
Worsley, S.F., Innocent, T.M., Heine, D., Murrell, J. Colin, Yu, D.W., Wilkinson, B., Hutchings, M.I., Boomsma, J.J. & Holmes, N.A. 2018: Symbiotic partnerships and their chemical interactins in the leafcutter ants (Hymenoptera: Formicidae). – Myrmecological News 27: 59-74.




Recent Comments